The 1,3‐Dipolar Cycloaddition: From Conception to Quantum Chemical
Par un écrivain mystérieux
Description

PDF) Recent advances in the photochemistry of the carbon-nitrogen double bond
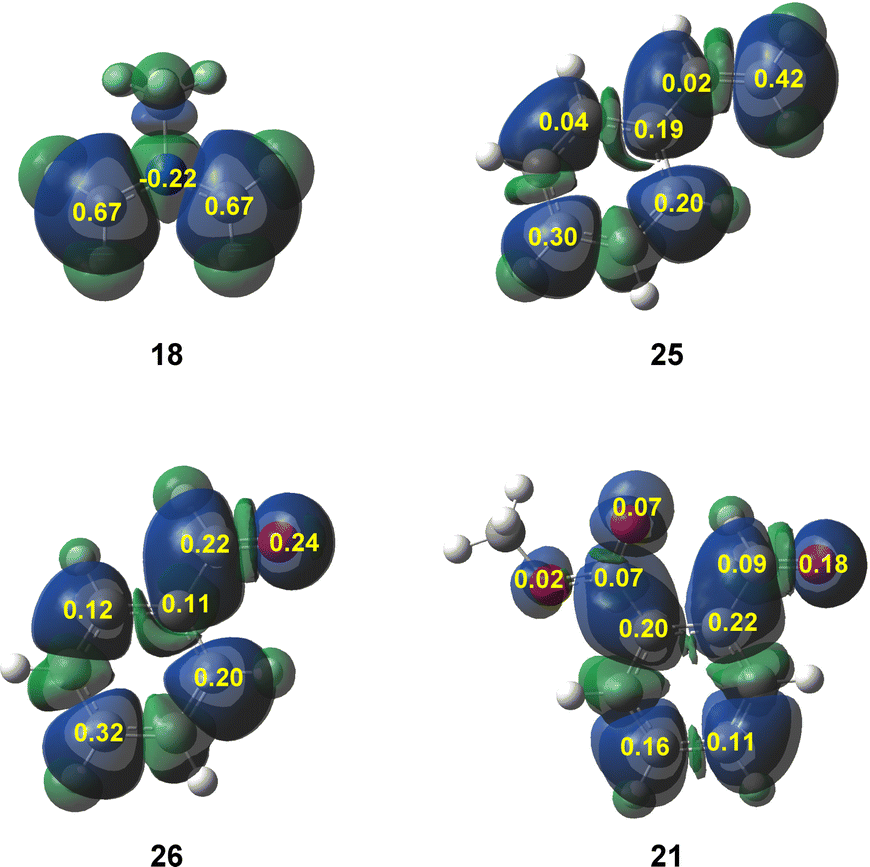
Unveiling the high reactivity of experimental pseudodiradical azomethine ylides within molecular electron density theory - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/D2CP05032C
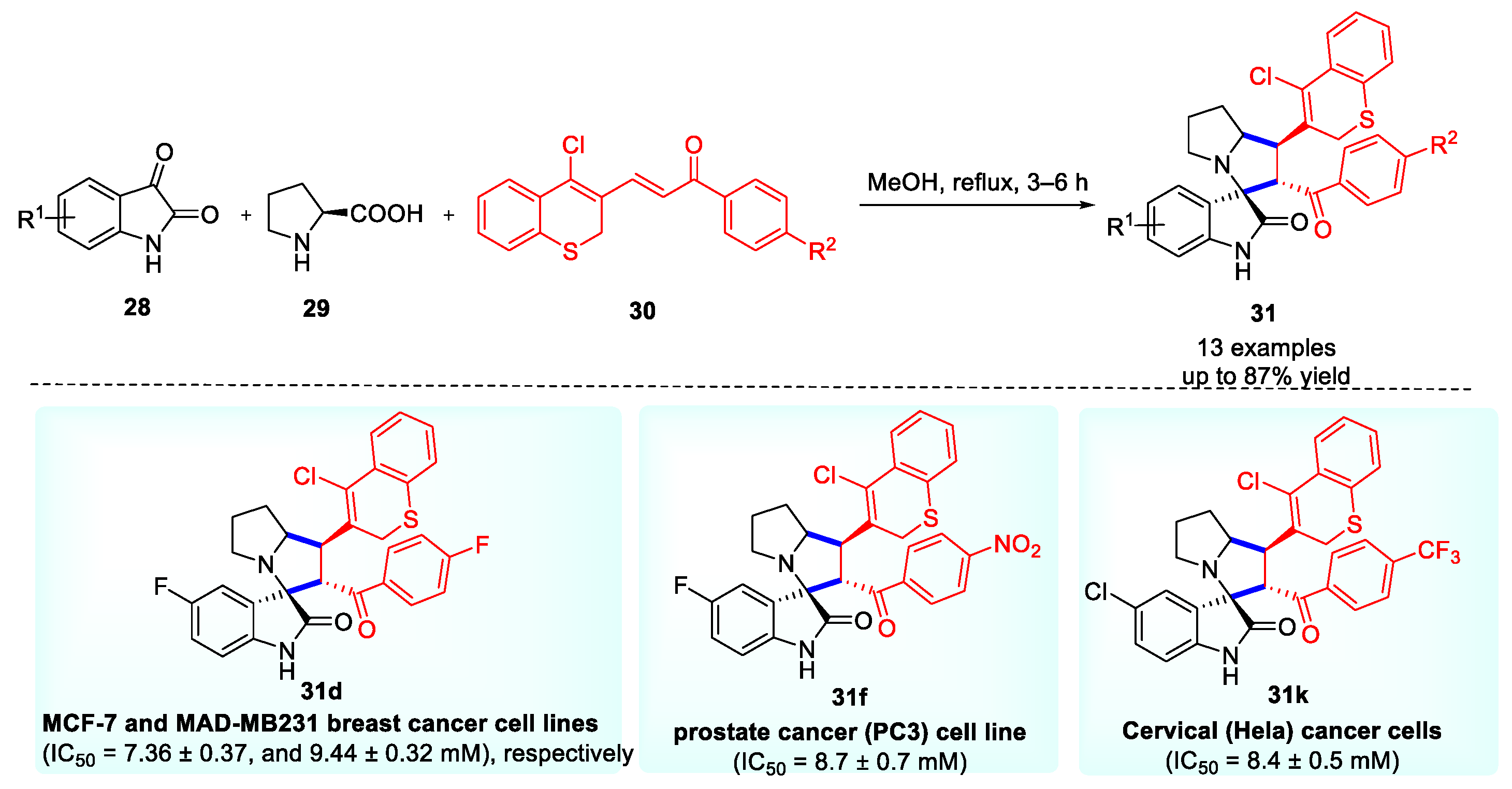
Molecules, Free Full-Text

Diradical-singlet character of 1,3-dipoles affects reactivity of 1,3-dipolar cycloaddition reactions and intramolecular cyclization

Approach to a Substituted Heptamethine Cyanine Chain by the Ring Opening of Zincke Salts.
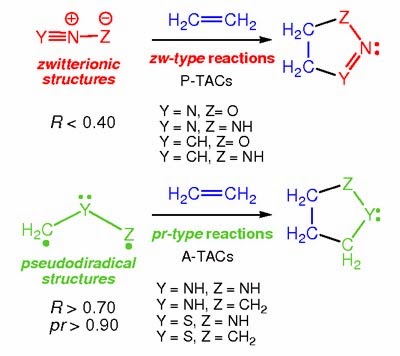
Molecular Electron Density Theory (MEDT) : Comments

Molecules, Free Full-Text
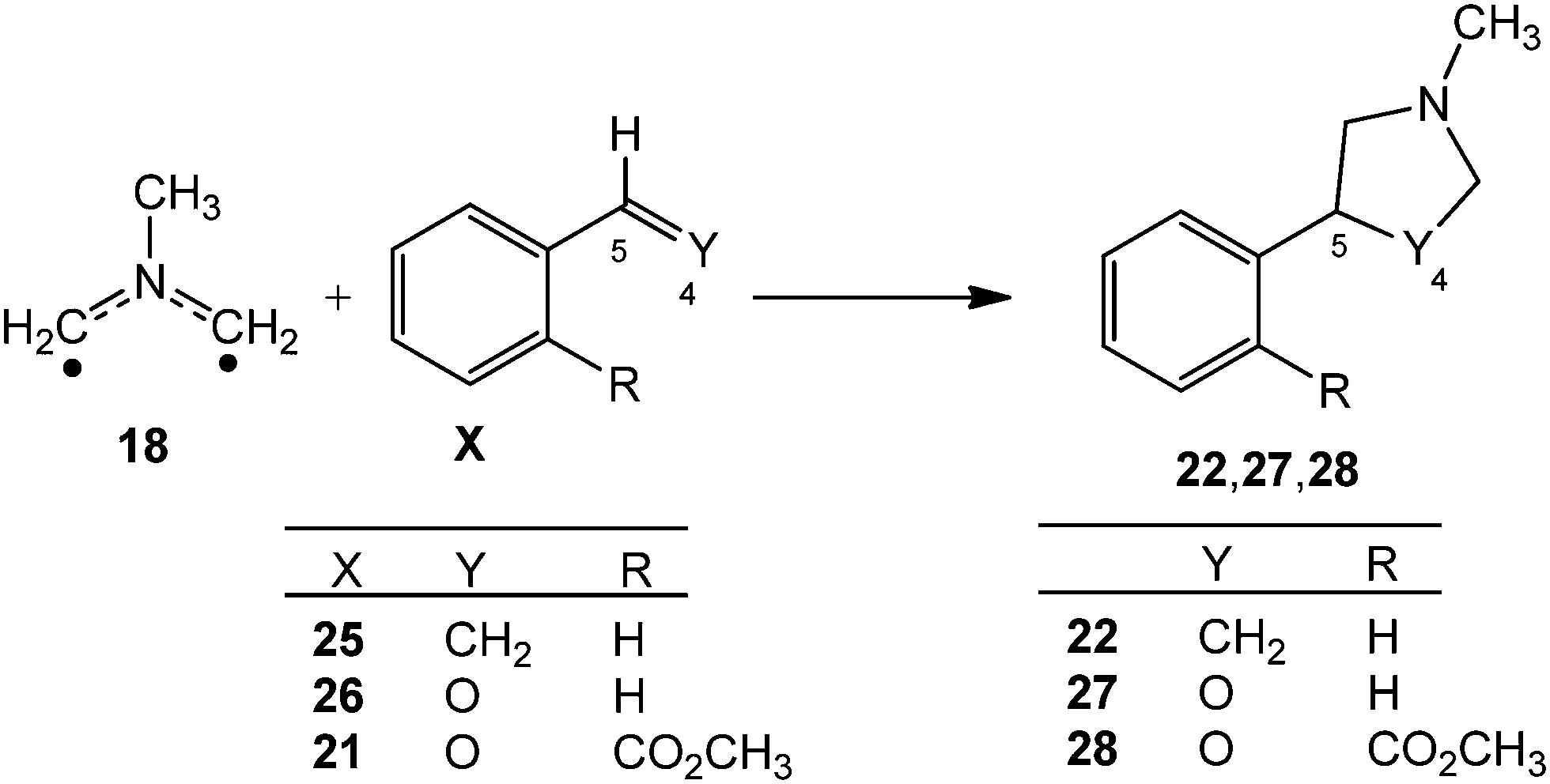
Unveiling the high reactivity of experimental pseudodiradical azomethine ylides within molecular electron density theory - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/D2CP05032C
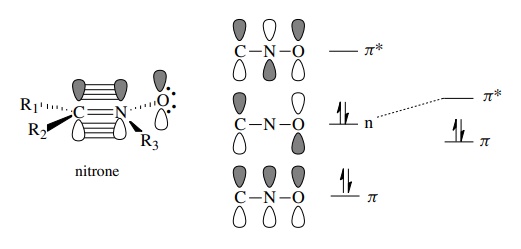
1,3-Dipolar Cycloadditions - Planning Organic Syntheses

BJOC - Search Results
Buy Trimethyl cyclopropane-1,2,3-tricarboxylate, 717-69-1

Fine-Tuning Strain and Electronic Activation of Strain-Promoted 1,3-Dipolar Cycloadditions with Endocyclic Sulfamates in SNO-OCTs. - Abstract - Europe PMC

The 1,3‐Dipolar Cycloaddition: From Conception to Quantum Chemical Design - Beutick - 2022 - Chemistry – An Asian Journal - Wiley Online Library

1, 3-Dipolar cycloaddition reactions of selected 1,3-dipoles with 7-isopropylidenenorbornadiene and follow-up thermolytic cleavage: A computational study - ScienceDirect
depuis
par adulte (le prix varie selon la taille du groupe)